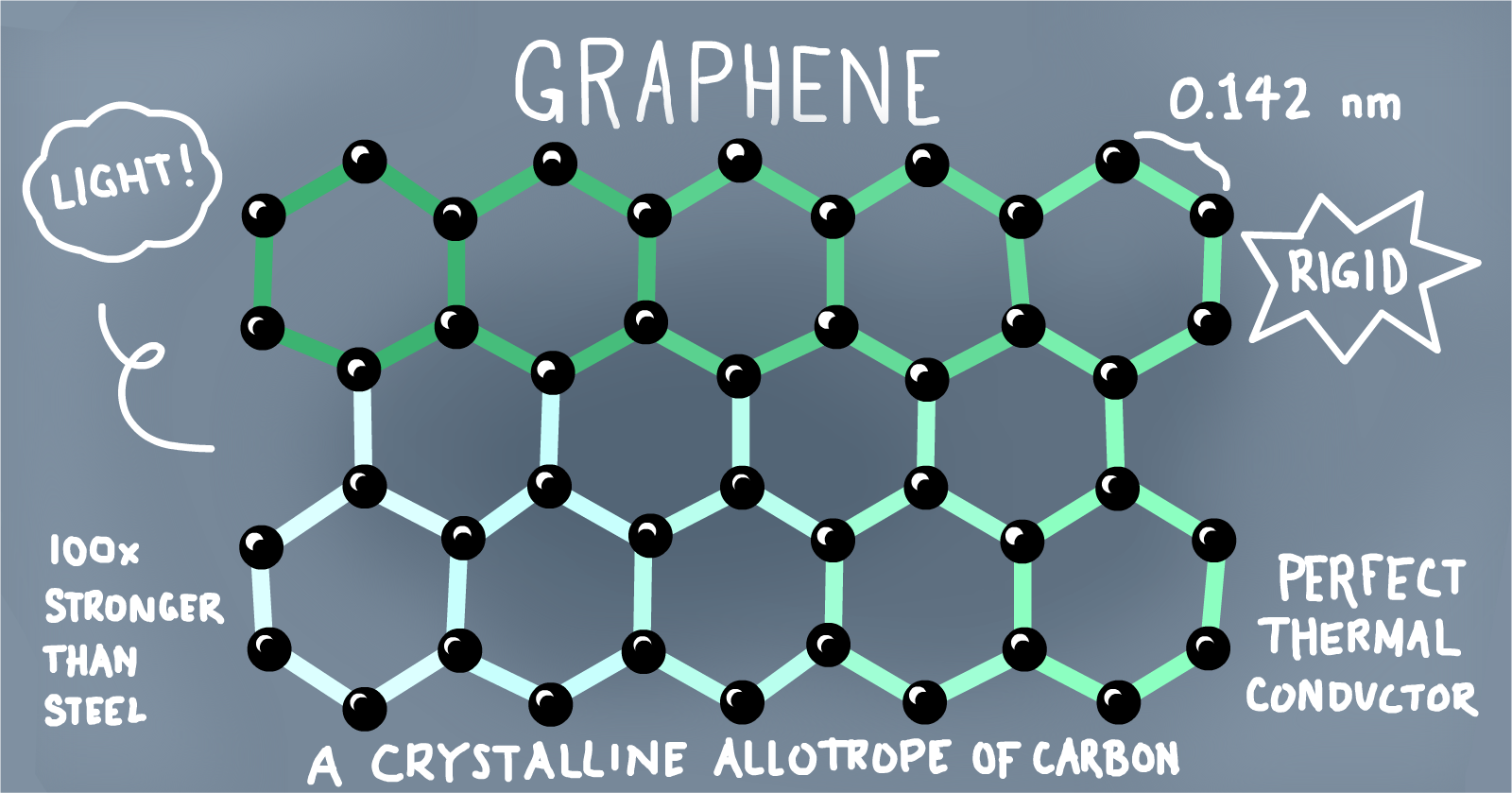
Graphene is probably one of the most talked about nanomaterials of the 21st century. As fate would have it, the core of number 2 pencils was meant to be much more than just for bubbling scantron sheets.
“We fooled nature by first making a three-dimensional material, which is graphite, and then pulling an individual layer out of it” — Dr. Andre Geim, inventor of graphene
In 2003, Dr. Andre Geim struck upon the idea of extracting thin, two-dimensional sheets from a graphite block using strong and sticky Scotch tape. Much to the founder’s surprise, the resulting thin flakes proved themselves to be a worthy discovery; graphene’s electrons are able to move close to the speed of light, carrying information 100 times faster; to put that into perspective, a graphene-charger could give our phones a full boost within a matter of seconds. Additionally, the material is durable and lightweight, with transparent properties suitable for optical applications.
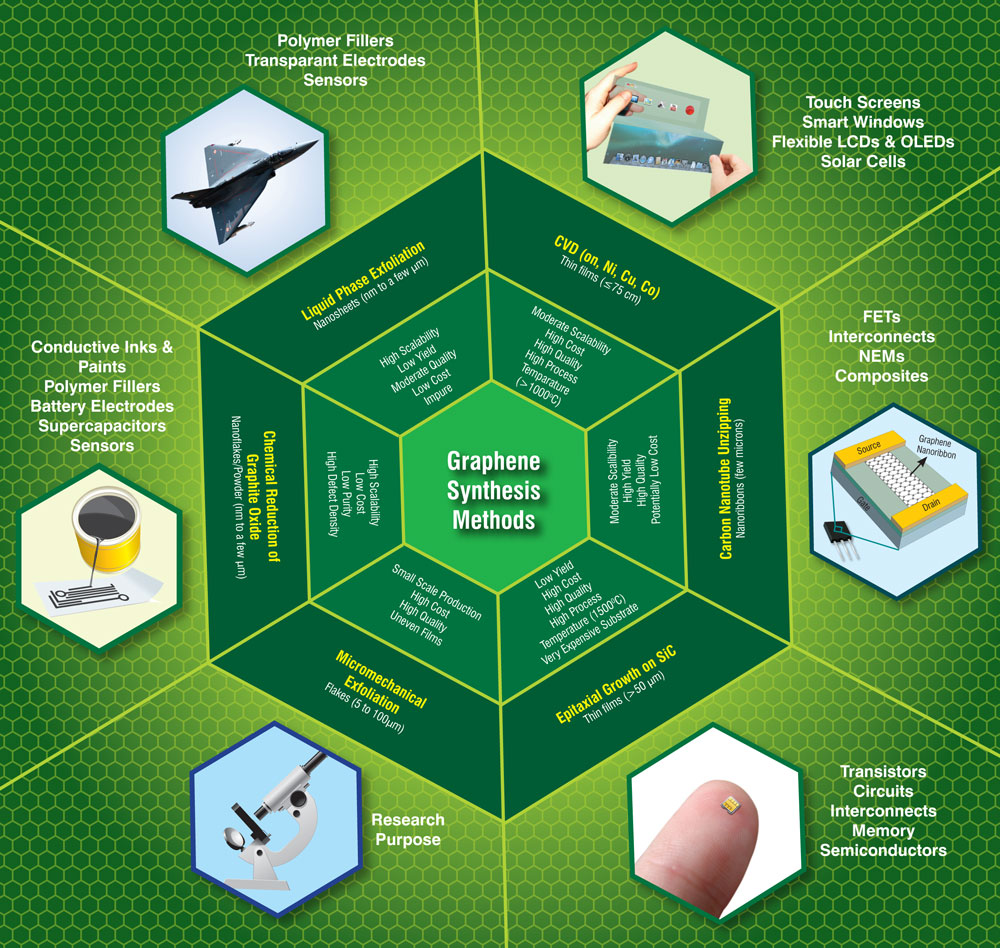
The perfect structure of graphene makes it a highly-researched material, but while scotch tape may be sufficient for the initial run, mass production requires more robust, scalable, and consistent manufacturing methods. Since the material’s discovery, graphene production has had more than a decade to advance, with a few established methods in the lead.
photo credit: nanowerk
According to Nanowerk’s in-depth report of global manufacturing patents, the most patented graphene production methods include CVD and exfoliation, followed by a trail of other methods including epitaxial growth and chemical synthesis. But, as the report further investigates, most of these methods require utmost care in handling: “The synthesis of graphene by conventional methods involves the use of toxic chemicals and these methods usually result in the generation hazardous waste and poisonous gases.”
While graphene’s superpower abilities can potentially yield unprecedented technological advancements, long-term chemical effects could tie it down to a sea of toxic residue, to be faced sooner or later. At a time where industrial global standards are rising, graphene production needs further improvement, even if the material is a “miracle.” To this end, university researchers are offering an alternative environmentally-friendly, one-step graphene production method via an ultra-bright lamp ablation; it requires no catalysts or toxic chemicals — only pure graphite, light, and heat — giving the process a clean upgrade.
Leveraging optics for graphene production
Australia and Israel have always been at the forefront of renewable advancements, as evident by the two country’s extensive list of renewable energy projects (here and here). The latest collaboration effort from scientists at the University of Western Australia and Ben-Gurion University is definitely in-line with those endeavors. As recently published in a Carbon paper, the researchers have proved that high-temperature lighting is actually all it takes to turn graphite flakes into graphene nanofilms, with nothing fancy (or toxic) required — except for a neat optical device.
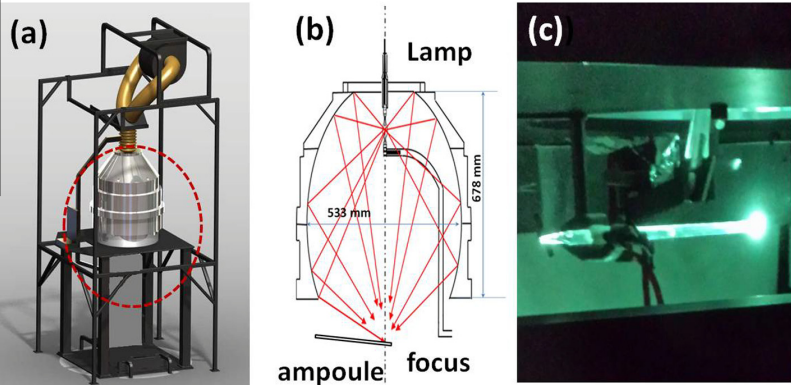
lamp ablation model and schematic
A magic lamp… ? Developed by Professor Jeffrey Gordon at Ben-Gurion University’s Solar Energy and Environmental Physics Department, the lamp had already proved its ability in developing silicon nanowires just a couple of years prior. And now they’ve discovered that a similar tactic can be used to make ultra-thin graphene films.
The mechanism behind the optical device is actually quite simple; the lamp creates an ultra-hot reactor environment of 2000-3000 K and produces a continuous irradiation by a sharp flux. The flux weakens the van der Waals interactions between graphite and brings it to a gaseous state. Then as the product moves out of the hot zone and into a substantially cooler state, it “quenches” and reforms into a 4-5 layer graphene film, measuring 1-2 nanometers in thickness. The entire process takes a little under an hour and results in a high-yield (~50%) film with spectroscopic characteristics, dimensions, and surface properties comparable to those currently being fabricated through chemical means.
One thing’s for sure, their lamp method makes conventional synthesis look overly-complicated and unnecessary. This is the shortcut researchers have been looking for in terms of time, cost, and material.
According to the team: “The [lamp ablation] process is relatively faster, safer and green — devoid of any toxic substances — just graphite plus concentrated light.”
Following their success, Professor Gordon shares they are now looking to further improve the underlying technology: “Much research is now on the agenda. That research will relate to several key issues, such as the size of graphene islets, the degree of uniformity in their size distribution, the yields, and how several possible high-volume synthesis methods affect them.” Specifically, they’re looking at ways to modify the lamp’s reactor geometry and irradiation distribution to scale-up production and increase quality assurance of the resultant film.
So does this mean we can finally look forward those cool graphene technologies that are making headlines?
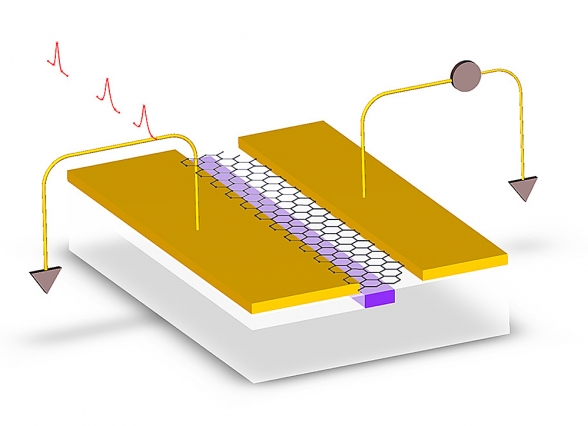
— Graphene photodetectors proposed by MIT researchers, which will make it possible for us to ditch electrons and transfer information through photons at the speed of light:
photo credit: mit.edu
— Color-tunable graphene LED lights proposed by Tsinghua University researchers, making it possible for one LED to emit an entire array of spectrum by varying the voltage:
photo credit: nature.com
— Stretchable “kirigami” graphene transistors proposed by Cornell physicists, making flexible nanodevices possible, even in the shape of a crane:
photo credit: cornell.edu
— Graphene ultrasonic radios proposed by physicists at the University of California, Berkeley, allowing humans to create our own version of bat-like and dolphin-like echolocation:
photo credit: Zettl Research Group
Probably, graphene still has a way to go before reaching widespread commercialization, as much of its development is still in infancy stage. However, the new advantageous procedure for synthesizing the few-layer nano-film can make the material more accessible at lower costs, says Professor Gordon. That’s one more obstacle down in bringing this miracle film out of the lab and into the world.
In the words of Andre Geim himself: “Introducing a new experimental system is generally more rewarding than trying to find new phenomena within crowded areas.” Creating graphene using only the parent material and concentrated light is indeed inventive and daring — and we hope this team’s research will lead to rewarding results further down the road.
Paper cited: Synthesis of few-layer graphene by lamp ablation